AR SIGNALING
INHIBITOR RESISTANCE
There are multiple mechanisms driving resistance to FDA approved Androgen Receptor Signaling Inhibitors (ARSi), such as Zytiga® (Abiraterone Acetate) and Xtandi® (Enzalutamide). Numerous novel therapeutics are being developed for metastatic castration resistance prostate cancer (mCRPC) to target these specific mechanisms of resistance which may be exploited for the development of therapy selection devices or utilized to demonstrate therapeutic pharmacodynamics and efficacy during the drug development process.
At Epic Sciences, we work with leading academic and government investigators to develop strategies for identifying resistance mechanisms and creating non-invasive tests that aid in the selection and monitoring of patients during clinical trials. These include tests to measure:

Tumor heterogeneity
Valuable in the clinical development of targeted therapy and combination therapyLearn more (below)
AR/PI3K reciprocal feedback
Valuable in the development of PI3K/AKT inhibitorsLearn more (below)
1. AR/PTEN Loss
AR independence
Valuable in design of clinical trials and measures of resistanceLearn more (below)
2. PSMA
AR-V7: Measure of nuclear localized AR-V7 derived truncated protein in CTCs
The measure of nuclear AR-V7 protein is a treatment specific biomarker in mCRPC that demonstrates therapy interaction and supports clinical utility. Fig.1 highlights the poor overall survival rate (HR=11.62) of patients who are biomarker positive for the Epic Sciences AR-V7 test and the 76% reduction of risk of death the use of taxane chemotherapy.
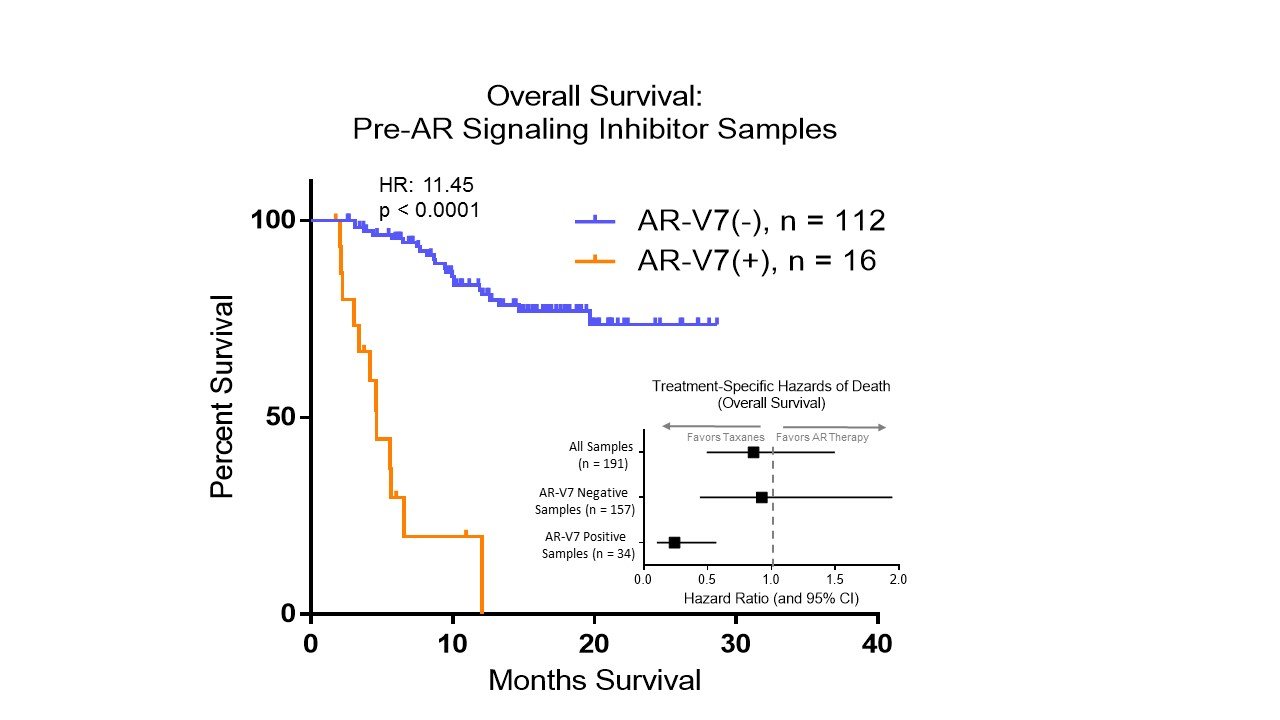
Figure 1. KM Curve of pre-therapy AR-V7 status: AR-V7 positive patients show resistance to AR Signaling Inhibitors and thus exhibit shorter overall survival.
GR CTC ANALYSIS
Glucocorticoid Receptor (GR) upregulation is a known resistance mechanism to ARSi, in which GR upregulation and binding bypasses ARSi. Activated GR proteins can bind to Androgen Response Elements (ARE) and enabling AR signaling despite effective ARSi activity. We have developed a GR CTC test to bind to upregulated GR. In a recent study, GR+ CTCs associated with worse OS in mCRPC patients and may help to identify patients who may benefit from novel GR inhibitor therapy. ASCO GU 2017 Poster
AR/PTEN CTC Analysis
AR resistance can occur through reciprocal feedback between PI3K and AR signaling. To measure cooperative AR/PI3K signaling we have developed single CTC phenotype and genotypic tools to assess AR protein expression, AR amplification and PTEN deletions within single CTCs.
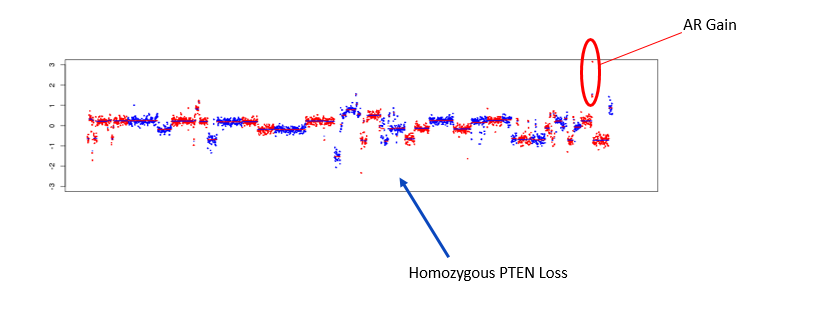
Figure 3. Copy Number Variation plot of a single CTC from a mCRPC patient expressing homozygous PTEN loss in conjunction with AR gain.
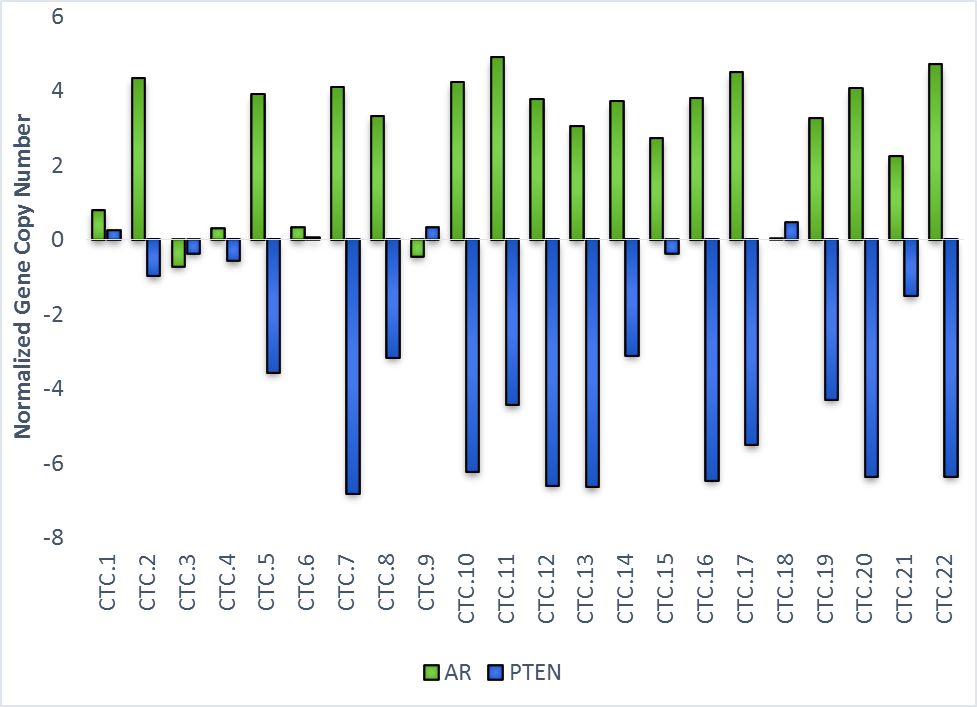
Figure 4. NextGen sequencing results of mCRPC patients indicate variety of single CTCs that exhibit coinciding AR amplification and PTEN deletion
CTC Phenotypic Heterogeneity
A subset of mCRPC patients harbor high tumor heterogeneity, which can occur at any line of therapy. NGS profiling of single tumor cells has demonstrated that these patients have polyclonal disease in which multiple drivers of disease are present in subclonal populations. The presence of high tumor heterogeneity thwarts narrowly, targeted therapeutic approach, even in cases where driver alterations of the targeted therapy are present. To assess this phenomenon, high resolution digital pathology was used to identify patients with a high degree of phenotypic heterogeneity in their circulating tumor cells. In a characterization of 179 patients with mCRPC, published in Cancer Research, patient blood samples were analyzed prior to initiating ARSi or taxane therapies to determine if CTC heterogeneity predicted therapy. Multivariate analyses revealed a statistically significant therapy interaction between increasing CTC heterogeneity (measured by the Shannon diversity index), measuring and higher risk of death on ARSi relative to taxanes.
Neuroendocrine Prostate Cancer (NEPC) CTC Classifier
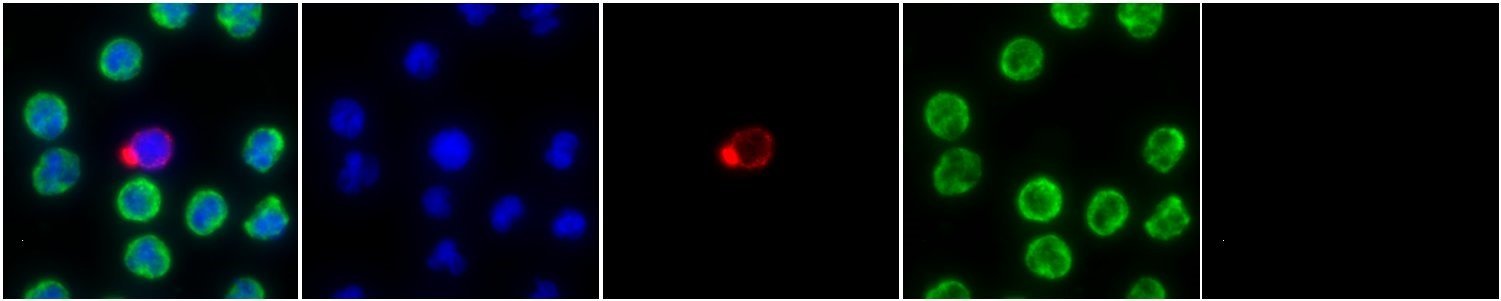
CTCs from patients with NEPC determined through metastatic biopsy that confirmes Small Cell Prostate Cancer Pathology or NE expressing tumor cells have a unique morphologic phenotype. While analysis of this cell type is specific to NEPC patients, ~10% of mCRPC patients have sub-clonal populations of NEPC CTCs. These patients demonstrate a worse response to AR inhibitors and are more likely to harbor visceral metastases. Read the paper here.
Figure 6. Cell image of a “Small CTC” exhibiting small morphology and positive for Cytokeratin expression. Prevalence of these cells are indicative of neuroendocrine disease.
PSMA
Our mission is to improve patient lives by enabling precision medicine with non-invasive tests that profile rare cells in cancer.
UP TO DATE WITH EPIC SCIENCES
JUST RELEASED: AR-V7 as an indication for treatment selection
